Microblog
A Systematic Identification of Pan-Genome Effector Families Responsible for Inducing Plant Immunity
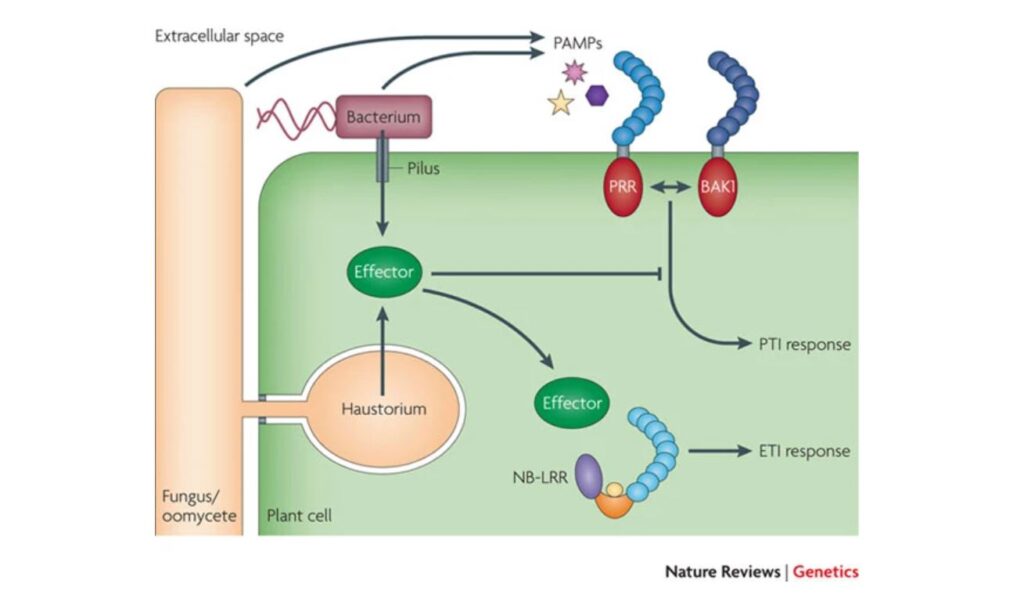
Plant pathogens represent a major threat to global ecosystems and the agricultural industry. Similar to all microbial niches, plant pathogens must adapt to their environment, including navigating the geographical range and immune defense of their host (Jones & Dangl, 2006). Characterizing the mechanisms underlying host-pathogen interactions are crucial for the development of engineered, broadly resistant crops. Many host-pathogen interactions are characterized by the arms race between the virulence of a given pathogen and the specificity of the host immune response. In plant-pathogen interactions, pathogens can suppress the first line of plant immune defenses and facilitate their own growth through the direct injection of effector proteins (Cui et al. 2009). In response, plants have co-evolved a second layer of defense that can identify these injected proteins and mount a specific immune response. This is termed effector-triggered immunity (ETI) and involves detection of effector proteins by the host’s intracellular nucleotide-binding leucine-rich repeat (NLR) receptor proteins (Qi & Innes, 2013). Previously, it was understood that the first line of plant immune defense involved broad-spectrum pathogen recognition and responses (Cui et al., 2015). In contrast, ETI was thought to be limited to the protection of specific plant genotypes from specific pathogens (Cui et al., 2015). Laflamme and colleagues (Science, 2020) demonstrated that the specificity of these interactions may have broader implications than previously suggested. Through a systematic analysis of 70 effector families identified across ~500 strains of Pseudomonas syringae, the authors determined that 96.8% percent of strains included at least one effector capable of inducing an ETI response in the plant, Arabidopsis thaliana.
To better understand ETI in A. thaliana, Laflamme and colleagues used computational methods to query the genomes of 494 P. syringae strains isolated from over 100 different plant species for putative effector proteins. This resulted in the identification and subsequent synthesis of 529 effectors in an expression plasmid library. Each plasmid included a single representative effector of one of the clades spanning 70 homology families. This library was transformed into P. syringae and used to systematically infect A. thaliana. As indicated by a reduction in discoloured plant tissue, the authors identified effectors that were able to induce the ETI response. Out of 529 effectors, 59 were able to robustly elicit the ETI response in A. thaliana. Surprisingly, of the 494 P. syringae strains examined in this paper, 478 (97%) contained at least one homolog of an ETI-eliciting effector. This indicated that the vast majority of P. syringae strains have the capability of eliciting an ETI response; however, these responses could be influenced by meta-effector interactions, expression on chromosomes versus plasmids and amino acid divergence between homologs.
To better understand which NLR genes are important for ETI induction, the authors infected a collection of A. thaliana mutants lacking different NLR proteins with P. syringae to identify loss-of-ETI mutants. Based on their results, the authors predicted that eight NLRs would be able to recognize close to 97% of P. syringe strains based on the presence of effector homologs. In fact, two NLR genes identified in the screen, ZAR1 and CAR1, could potentially identify and induce ETI in response to close to 95% of P. syringe strains.
Collectively, Laflamme and colleagues identified a core set of P. syringae effectors important for eliciting ETI and determined that the host ETI response converges on only a few NLRs. Overall, this work identifies a key set of plant genes important for the broad response to pathogens. Continued investigation to dissect the mechanisms underlying host-specificity and pathogen virulence will be necessary to engineer broadly resistant crops and defend against the threat plant pathogens pose to ecosystem health and global food security.
Primary Research Article
Laflamme B, et al. The pan-genome effector-triggered immunity landscape of a host-pathogen interaction. Science 367, 763-768 (2020).
References
Jones JDG, Dangl JL. The plant immune system. Nature 444, 323-329 (2006).
Cui H, Xiang T, Zhou J-M. Plant immunity: a lesson from pathogenic bacterial effector proteins. Cell Microbiol 11, 1453-1461 (2009).
Qi D, Innes RW. Recent advances in plant NLR structure, function, localization, and signaling. Front Immunol 4, 1-10.
Cui H, Tsuda K, Parker JE. Effector-triggered immunity: From pathogen perception to robust defence. Ann Rev Plant Biol 66, 487-511 (2015).