Microblog
New Perspective on Antiretroviral Treatments: Restricting HIV-1 Replication by Targeting Intracellular Signaling Pathways
 Human immunodeficiency virus 1 (HIV-1) is a sexually transmitted retrovirus that attacks the immune cells of the body and can lead to acquired immunodeficiency disease (AIDS). HIV-1 is composed of an RNA genome that becomes integrated into the human genome post-infection and encodes at least 18 viral proteins that are expressed from alternatively spliced forms of the RNA sequence. With 38 million people infected with HIV-1 globally and no existing cure, strategies to control this viral infection are a major priority (amfAR 2019). The current gold standard treatment for HIV, called antiretroviral therapy (ART), entails administration of a combination of drugs that reduce the viral load, which prolongs life and limits further spread of the virus (CDC 2019). Unfortunately, ARTs have led to a substantial rise in drug resistant strains of HIV-1, promoting treatment failure and highlighting the need for alternative approaches to eradicate this disease. To address this problem, Wong et al. (2020) explored a novel strategy to target HIV-1 infection.
Human immunodeficiency virus 1 (HIV-1) is a sexually transmitted retrovirus that attacks the immune cells of the body and can lead to acquired immunodeficiency disease (AIDS). HIV-1 is composed of an RNA genome that becomes integrated into the human genome post-infection and encodes at least 18 viral proteins that are expressed from alternatively spliced forms of the RNA sequence. With 38 million people infected with HIV-1 globally and no existing cure, strategies to control this viral infection are a major priority (amfAR 2019). The current gold standard treatment for HIV, called antiretroviral therapy (ART), entails administration of a combination of drugs that reduce the viral load, which prolongs life and limits further spread of the virus (CDC 2019). Unfortunately, ARTs have led to a substantial rise in drug resistant strains of HIV-1, promoting treatment failure and highlighting the need for alternative approaches to eradicate this disease. To address this problem, Wong et al. (2020) explored a novel strategy to target HIV-1 infection.
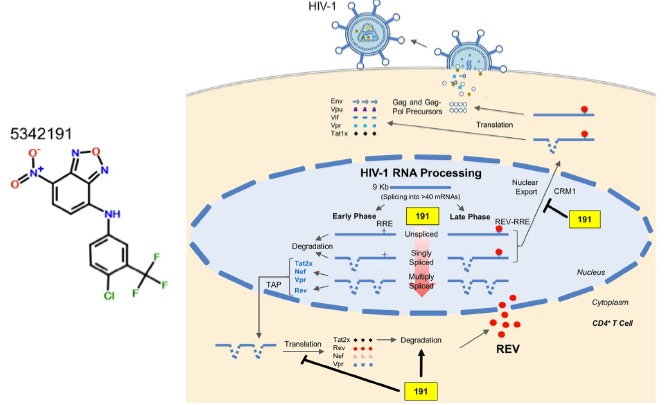
To uncover alternate methods of HIV-1 restriction, Wong et al. screened more than 60 compounds for their effect on HIV-1 replication post-integration. Using a human cell line containing integrated HIV-1 genome, the authors identified compound 5342191, a benzoxadiazole, which significantly inhibited viral gene expression in a dose-dependent manner, with no effect on the viability of human cells. In particular, 5342191 reduced expression of several essential structural proteins and regulatory factors (including those involved in RNA transcription and nuclear export), which are untargeted by any currently available HIV-1 inhibitors. Furthermore, 5342191 treatment of CD4+ T cells, the primary cell type infected by HIV-1, inhibited replication of both wild-type and drug-resistant strains of the virus, demonstrating the efficacy of this molecule.
To gain insights into how 5342191 restricts infection, the authors performed RNA analysis and found 5342191 affects viral RNA accumulation. Specifically, levels of unspliced or singly-spliced mRNAs were depleted while multiply-spliced mRNA levels increased post-treatment. These changes were attributed to the altered abundance of cellular splicing factors including specific SR proteins. The defect in unspliced mRNA accumulation was complemented with a decrease in translocation of viral mRNAs to the cytoplasm, consistent with loss of the HIV-1 nuclear export factor, Rev. In contrast to affecting viral RNAs, sequencing analysis showed that 5342191 had minimal effects on the level of global cellular transcripts, inducing changes to only a small subset of cellular RNAs, highlighting the selectivity of this inhibitor. Although multiply-spliced viral RNAs increased while Tat, a viral factor involved in activating HIV-1 transcription, decreased in expression upon drug treatment, this was not due to perturbation in de novo synthesis of Tat but rather due to higher degradation of the protein. Similar to cellular RNA levels, 5342191 treatment had little effect on the host proteome.
Next, the authors shed light on the cellular mechanisms by which 5342191 inhibits HIV-1 gene expression by comparing 5342191 to another drug type called cardiotonic steroids (CSs). CSs alter HIV-1 RNA processing partly through activation of the mitogen-activated protein kinase (MAPK) pathway. Through this comparison, the authors revealed that while 5342191 utilizes the same signaling pathway, its exact mechanism of action is different from CSs. This was because 5342191 did not have any effect on other intermediary pathway molecules as CSs. Follow-up experiments found that inhibition of the MEK1/2 components of the MAPK pathway rescued HIV-1 gene expression in 5342191-treated cells, confirming the involvement of the this pathway in regulating viral gene expression. The authors also observed that 5342191 treatment increased Ras GTPase activity in cells, giving rise to the hypothesis that the Ras pathway might be activated upstream of the MAPK pathway to restrict HIV-1 replication. Experiments found that by inhibiting signaling cues by the G-protein coupled receptors (GPCRs) that normally activate the Ras pathway, HIV-1 gene expression was restored and the viral mRNA changes previously observed in the presence of 5342191were reversed. This validated that 5342191 works by activating the GPCR signaling in cells to restrict HIV-1 replication.
Overall, this study provides a novel mechanism of HIV-1 restriction through activation of an intracellular signaling pathway. The authors identify that compound 5342191 works differently than currently used drugs which are limited in their usage due to either their toxicity or high chance of HIV-1 developing resistance to them. 5342191 not only inhibited HIV-1 gene expression of wild-type strains but also drug-resistant strains of the virus in infected cells, making it a promising inhibitor for development into a future HIV-1 therapy.
Primary Reference:
Wong, R.W., Balachandran, A., Cheung, P.K., Cheng, R., Pan, Q., Stoilov, P., Harrigan, P.R., Blencowe, B.J., Branch, D.R. and Cochrane, A., 2020. An activator of G protein-coupled receptor and MEK1/2-ERK1/2 signaling inhibits HIV-1 replication by altering viral RNA processing. PLoS pathogens, 16(2), p.e1008307.
Other References:
amfAR. Basic Facts about HIV/AIDS. https://www.amfar.org/facts-about-hiv-and-aids/#a6 (2019). Accessed July 14, 2020.
Centers for Disease Control and Prevention. HIV Treatment. https://www.cdc.gov/hiv/basics/livingwithhiv/treatment.html (2019). Accessed July 16, 2020.

Frappier Lab
Posted on : 11/06/2020 9:00 AM
Liked this post?
Click the social media icons on the left to share!
Have anything to say? Comment below!