Microblog
Evaluating the “Pro” of Probiotics: No Benefits of Lactobacillus Therapy for Pediatric Acute Gastroenteritis
 Over the past decade, the human nutrition and disease management industries have converged upon a golden goose: probiotics. These live microbes, mostly Lactobacillus bacteria, command a multibillion-dollar global market built around fermented foods and dietary supplements. Their wide range of purported uses are based on the principle that beneficial bacteria can be introduced into and thrive within the gut, preventing the expansion of other microorganisms that may be harmful to the body. In addition to promising intestinal balance for the health-oriented consumer, probiotics have become an increasingly popular alternative for combatting ailments without effective treatment options. The marketing claims are impressive– and rather ambiguous– but how does the clinical evidence measure up?
Over the past decade, the human nutrition and disease management industries have converged upon a golden goose: probiotics. These live microbes, mostly Lactobacillus bacteria, command a multibillion-dollar global market built around fermented foods and dietary supplements. Their wide range of purported uses are based on the principle that beneficial bacteria can be introduced into and thrive within the gut, preventing the expansion of other microorganisms that may be harmful to the body. In addition to promising intestinal balance for the health-oriented consumer, probiotics have become an increasingly popular alternative for combatting ailments without effective treatment options. The marketing claims are impressive– and rather ambiguous– but how does the clinical evidence measure up?
Although recommendations vary and are not accepted by many clinical experts, probiotic administration is often promoted for certain gastrointestinal illnesses such as necrotizing enterocolitis, antibiotic-associated diarrhea and acute gastroenteritis (AGE) (Kechagia et al. 2013, Vecchio et al., 2016). However, most clinical trials conducted to-date have been hampered by limitations including insufficient sample sizes (Parker et al., 2018). Moreover, a number of recent large-scale studies have found no advantage to probiotic usage for these indications (Allen et al. 2013, Costeloe et al. 2016, Freedman et al. 2018, Schnadower et al., 2018).
Notably, a nation-wide trial examining the effects of probiotic treatment for AGE in young children was reported in late 2018 (Freedman et al., 2018). The randomized, double-blind study included nearly 900 patients who presented to pediatric emergency departments with vomiting and diarrhea and subsequently received a clinical diagnosis of gastrointestinal infection. Implementation of an externally validated, composite measure scoring system and related secondary analyses determined that a 5-day combination probiotic course consisting of Lactobacillus rhamnosus R0011 and Lactobacillus helveticus R0052 did not reduce adverse outcomes compared to placebo. A concurrent study undertaken in the United States, which assessed the efficacy of L. rhamnosus GG, also obtained congruent results (Schnadower et al., 2018).
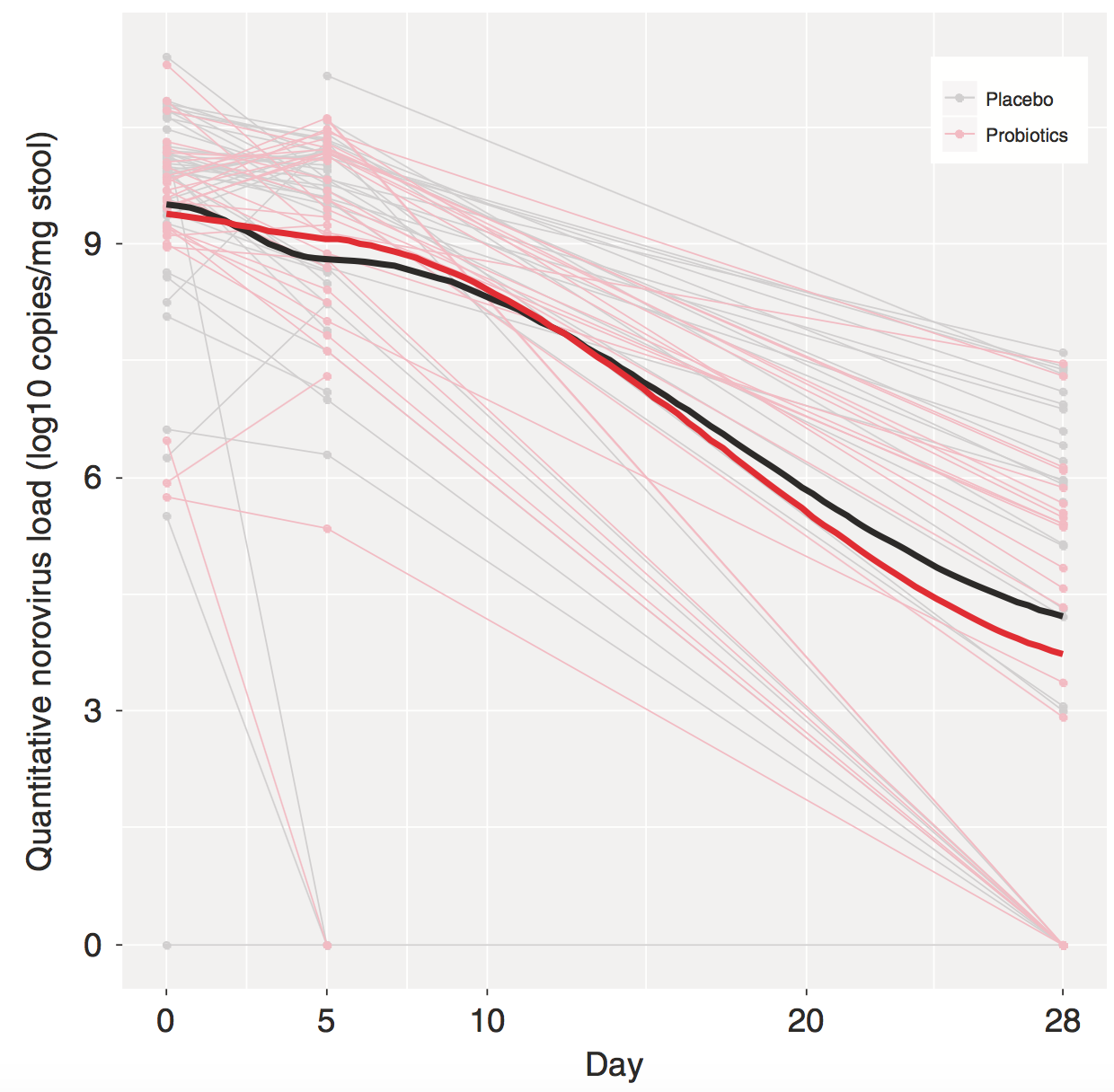
More recently, a follow-up analysis was performed to further explore these clinical data at the level of the causative pathogen (Freedman et al. 2020). Rotavirus is historically the leading cause of AGE worldwide, though the predominant etiological agent in North America has now shifted to norovirus following the introduction of a rotavirus vaccine (Payne et al., 2013). Previous work has suggested divergent effectiveness of probiotics against rotaviral- compared to noroviral-induced AGE (Szymański et al., 2006), particularly given the underlying differences in their pathogenic mechanisms (Navaneethan and Giannella, 2008). Consequently, the lack of response to probiotic treatment observed in the main study could have originated from these virus-specific determinants. In addition, noroviral load is known to correlate with AGE severity (He et al., 2017). As such, the researchers sought to quantify and compare the reduction in rotavirus, norovirus, as well as adenovirus load after probiotic administration. Real-time PCR using stool specimens collected at the time of initial study enrolment and two later time points enabled detection of common viral, bacterial, and parasitic enteropathogens. A viral pathogen was identified in 55.3% of patients, with no pathogen present in the next largest sub-cohort (36.6%). There were no statistically significant differences in clinical severity between treatment and placebo arms for the rotavirus, norovirus or adenovirus subgroups. Furthermore, the combination probiotic did not accelerate clearance of viral nucleic acids from patient stool. These findings further support that probiotic use is unable to improve clinical outcomes in pediatric AGE, regardless of the causative viral agent. Nevertheless, given the variability of local host gut microbiomes and infectious agents (Yatsuneko et al., 2012), these conclusions may not be applicable in other therapeutic contexts nor to all probiotic products available on the market.
We are still a long way away from establishing the role of probiotics in our health routines. It remains unclear how we can best promote or recreate a healthy human microbiota; however, there is reason to be optimistic for the therapeutic future of probiotics as researchers continue unravelling the complex network of gut microbial interactions. Certainly, a deeper understanding of the influence of genetic and environmental factors, alongside strong supporting evidence from large and rigorous clinical trials, will be critical for translating this potential to the clinics.
Primary Research Article:
Freedman, S.B., Xie, J., Nettel-Aguirre, A., Pang, X.-L., Chui, L., Williamson-Urquhart, S., Schnadower, D., Schuh, S., Sherman, P.M., Lee, B.E., Gouin S., Farion K.J., Poonai N., Hurley K.F., Qiu Y., Ghandi B, Lloyd C., Finkelstein Y., on behalf of the Pediatric Emergency Research Canada Probiotic Regimen (PERC) for Outpatient Gastroenteritis Utility of Treatment (PROGUT) Trial Group (2020). A randomized trial evaluating virus-specific effects of a combination probiotic in children with acute gastroenteritis. Nat Commun 11.
Other References:
Allen, S.J., Wareham, K., Wang, D., Bradley, C., Hutchings, H., Harris, W., Dhar, A., Brown, H., Foden, A., Gravenor, M.B., et al. (2013). Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): a randomised, double-blind, placebo-controlled, multicentre trial. The Lancet 382, 1249–1257.
Costeloe, K., Hardy, P., Juszczak, E., Wilks, M., Millar, M.R., and Probiotics in Preterm Infants Study Collaborative Group (2016). Bifidobacterium breve BBG-001 in very preterm infants: a randomised controlled phase 3 trial. Lancet 387, 649–660.
Freedman, S.B., Williamson-Urquhart, S., Farion, K.J., Gouin, S., Willan, A.R., Poonai, N., Hurley, K., Sherman, P.M., Finkelstein, Y., Lee, B.E., et al. (2018). Multicenter Trial of a Combination Probiotic for Children with Gastroenteritis. N. Engl. J. Med. 379, 2015–2026.
He, T., McMillen, T.A., Qiu, Y., Chen, L.H., Lu, X., Pang, X.-L., Kamboj, M., and Tang, Y.-W. (2017). Norovirus Loads in Stool Specimens of Cancer Patients with Norovirus Gastroenteritis. J Mol Diagn 19, 836–842.
Kechagia, M., Basoulis, D., Konstantopoulou, S., Dimitriadi, D., Gyftopoulou, K., Skarmoutsou, N., and Fakiri, E.M. (2013). Health Benefits of Probiotics: A Review. ISRN Nutr 2013.
Navaneethan, U., and Giannella, R.A. (2008). Mechanisms of infectious diarrhea. Nat Clin Pract Gastroenterol Hepatol 5, 637–647.
Parker, E.A., Roy, T., D’Adamo, C.R., and Wieland, L.S. (2018). Probiotics and Gastrointestinal Conditions: An Overview of Evidence from the Cochrane Collaboration. Nutrition 45, 125-134.e11.
Payne, D.C., Vinjé, J., Szilagyi, P.G., Edwards, K.M., Staat, M.A., Weinberg, G.A., Hall, C.B., Chappell, J., Bernstein, D.I., Curns, A.T., et al. (2013). Norovirus and Medically Attended Gastroenteritis in U.S. Children. New England Journal of Medicine 368, 1121–1130.
Schnadower, D., Tarr, P.I., Casper, T.C., Gorelick, M.H., Dean, J.M., O’Connell, K.J., Mahajan, P., Levine, A.C., Bhatt, S.R., Roskind, C.G., et al. (2018). Lactobacillus rhamnosus GG versus Placebo for Acute Gastroenteritis in Children. N. Engl. J. Med. 379, 2002–2014.
Szymański, H., Pejcz, J., Jawień, M., Chmielarczyk, A., Strus, M., and Heczko, P.B. (2006). Treatment of acute infectious diarrhoea in infants and children with a mixture of three Lactobacillus rhamnosus strains–a randomized, double-blind, placebo-controlled trial. Aliment. Pharmacol. Ther. 23, 247–253.
Vecchio, A.L., Dias, J.A., Berkley, J.A., Boey, C., Cohen, M.B., Cruchet, S., Liguoro, I., Lindo, E.S., Sandhu, B., Sherman, P., et al. (2016). Comparison of Recommendations in Clinical Practice Guidelines for Acute Gastroenteritis in Children. J Pediatr Gastroenterol Nutr 63, 226–235.
Yatsunenko, T., Rey, F.E., Manary, M.J., Trehan, I., Dominguez-Bello, M.G., Contreras, M., Magris, M., Hidalgo, G., Baldassano, R.N., Anokhin, A.P., et al. (2012). Human gut microbiome viewed across age and geography. Nature 486, 222–227.
Liked this post?
Click the social media icons on the left to share!
Have anything to say? Comment below!